The Evolution of Medicare Policy and the Strategic Implementation of GLP-1 Coverage Pilots
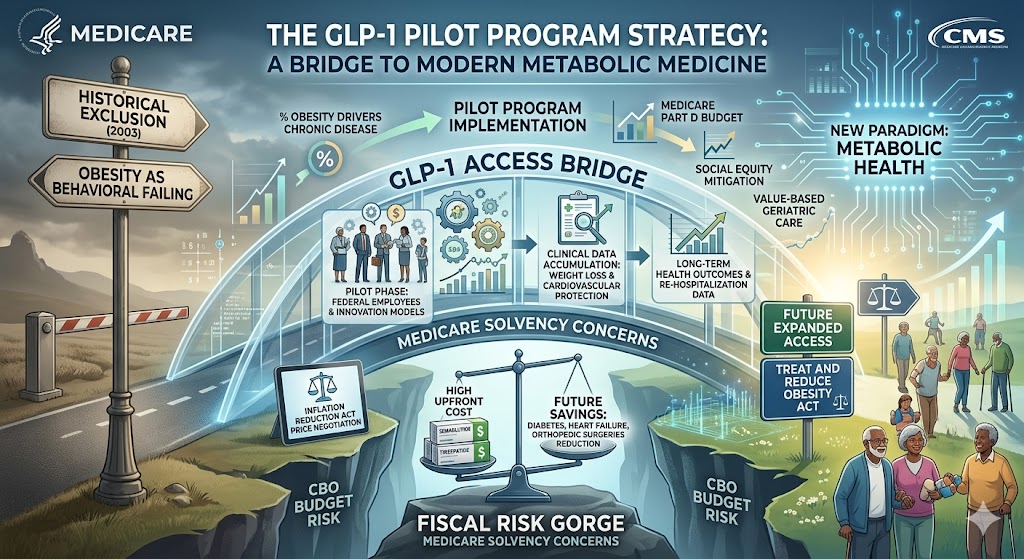
The landscape of federal healthcare coverage is currently undergoing a transformative shift as the Centers for Medicare and Medicaid Services (CMS) begins to navigate the complex intersection of obesity management and fiscal responsibility. Historically, Medicare has been prohibited by a 2003 statutory exclusion from covering medications used specifically for weight loss, a policy rooted in the era when such treatments were viewed primarily as cosmetic interventions. However, the emergence of glucagon-like peptide-1 (GLP-1) receptor agonists has fundamentally challenged this paradigm, forcing a re-evaluation of how the federal government categorizes and funds metabolic health treatments. The introduction of specific pilot programs signals a cautious yet definitive move toward acknowledging obesity as a primary driver of chronic disease rather than a behavioral failing.
The primary catalyst for this policy shift is the overwhelming clinical evidence demonstrating that GLP-1 medications, such as semaglutide and tirzepatide, do more than reduce body mass; they significantly lower the risk of major adverse cardiovascular events. Because Medicare is permitted to cover drugs that treat specific medical conditions, the FDA’s approval of these medications for reducing heart attack and stroke risks in patients with obesity opened a critical “backdoor” for coverage. This cause-and-effect relationship between clinical outcome data and regulatory flexibility has allowed CMS to broaden its scope, providing a pathway for beneficiaries with pre-existing heart disease to access these high-cost therapies under Medicare Part D.
A significant component of this evolution is the federal government’s pilot program targeting specific cohorts, including certain federal employees and participants in innovation models, to receive GLP-1s solely for weight loss. These pilots serve as a controlled environment to gather data on long-term health outcomes and fiscal impact. By monitoring a limited population, the government aims to determine if the high upfront cost of these medications is offset by a reduction in hospitalizations for diabetes, heart failure, and orthopedic surgeries. This data-driven approach is essential for the Congressional Budget Office (CBO), which currently views broad obesity drug coverage as a significant budgetary risk due to the sheer volume of eligible beneficiaries.
The economic tension surrounding GLP-1 coverage is a direct result of the discrepancy between drug pricing and the projected growth of the Medicare-eligible population. With list prices often exceeding one thousand dollars per month, the potential for these drugs to consume a disproportionate share of the Part D budget is high. This fiscal reality has led to a cautious implementation strategy where access is strictly tied to documented medical necessity. The government’s hesitation is not a reflection of the drugs’ efficacy, but rather a structural concern regarding the solvency of the Medicare Trust Fund in the face of a therapeutic class that could potentially be prescribed to tens of millions of seniors.
Furthermore, the pilot programs address the “cliff” effect of pharmaceutical intervention, where patients may regain weight if treatment is discontinued. By evaluating coverage within a pilot framework, federal health officials are investigating the sustainability of chronic obesity management. This involves analyzing whether intermittent dosing or long-term maintenance programs are more cost-effective. The success of these pilots will likely dictate the language of future legislation, such as the Treat and Reduce Obesity Act, which seeks to permanently remove the 1990s-era ban on weight loss drug coverage across all Medicare plans.
The impact of the Inflation Reduction Act (IRA) also plays a crucial role in this unfolding narrative. As GLP-1 medications eventually become eligible for price negotiations between the federal government and pharmaceutical manufacturers, the cost-benefit analysis for Medicare changes. Lowering the per-patient cost through federal negotiation will likely accelerate the transition from restricted pilot programs to broad, standardized coverage. This sequential progression—from clinical validation to pilot testing and finally to price-negotiated expansion—represents a systematic effort to modernize the Medicare benefit structure without triggering a fiscal crisis.
Social and health equity concerns are also driving the expansion of these pilot initiatives. Obesity disproportionately affects lower-income beneficiaries and minority populations who rely on Medicare and Medicaid for their primary care. By initiating pilot programs that include diverse demographic groups, the federal government is attempting to mitigate the risk of a “pharmaceutical divide” where only those with private supplemental insurance or significant personal wealth can access life-extending weight loss therapies. Consequently, these programs are being scrutinized for their ability to improve health outcomes in traditionally underserved communities, which could lead to substantial long-term savings for the public health system.
The role of physician and patient advocacy groups cannot be overlooked as a driver of these federal pilots. Professional medical organizations have been instrumental in reframing obesity as a chronic, relapsing disease state. This shift in medical consensus has exerted significant pressure on CMS to align its coverage policies with modern clinical guidelines. The resulting pilot programs are, in many ways, a compromise between the medical community’s demand for immediate access and the government’s requirement for fiscal predictability. This tension ensures that while coverage is expanding, it remains subject to rigorous prior authorization and clinical criteria.
Looking ahead, the implications of these pilot programs extend far beyond weight loss. They represent a fundamental change in the geriatric care model, moving from reactive treatment of complications to proactive management of metabolic health. If the pilots demonstrate a significant decline in the progression of comorbidities, Medicare may shift its entire reimbursement strategy to favor early pharmacological intervention for obesity. This would mark a departure from the traditional fee-for-service model and toward a value-based care system where the prevention of chronic illness is prioritized as a primary cost-saving measure.
The current state of Medicare coverage for GLP-1s remains a complex patchwork of conditional approvals and experimental pilots. Beneficiaries must navigate a system where eligibility is often determined by the presence of a secondary condition, such as Type 2 diabetes or cardiovascular disease, rather than obesity alone. This fragmentation is a direct consequence of the existing legislative ban, but the ongoing federal pilots are the most significant evidence to date that this ban is nearing its end. As the data from these programs matures, the federal government will be forced to make a definitive decision on the permanent inclusion of weight-loss medications in the Medicare formulary.
In conclusion, the federal government’s pilot programs for GLP-1 coverage are a strategic bridge between outdated statutory restrictions and the reality of modern metabolic medicine. The cause-and-effect loop—where clinical success leads to pilot testing, which in turn informs budgetary policy—is currently in its most critical phase. The outcomes of these initiatives will not only determine the health trajectory of millions of American seniors but will also set a global precedent for how nationalized health systems manage the financial and logistical challenges of the obesity epidemic. The transition from weight loss as a “lifestyle” issue to a “lifesaving” intervention is now a matter of federal policy in motion.