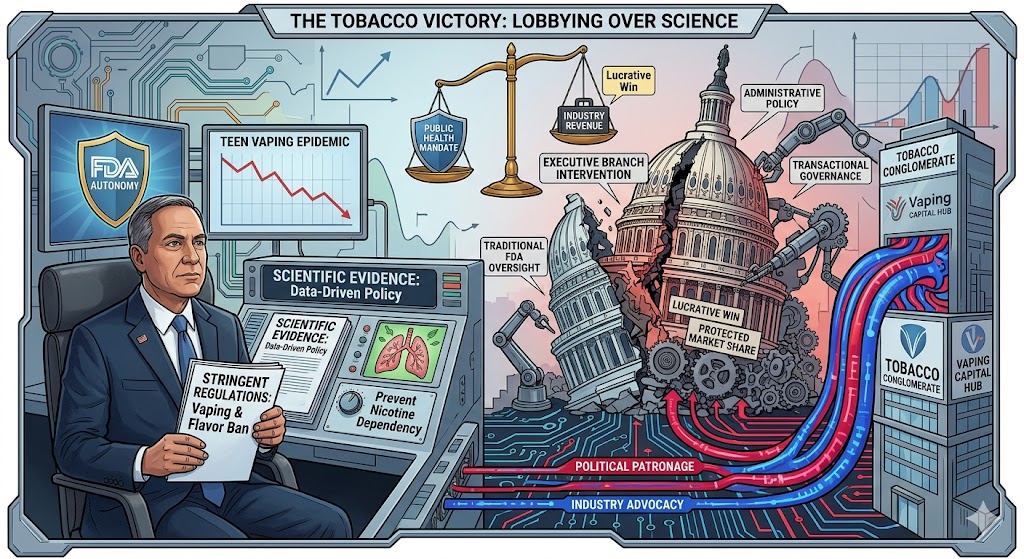
The Intersection of Political Patronage and Public Health Policy in the Tobacco Industry
The recent alignment between the executive branch and the tobacco industry represents a significant shift in the landscape of federal health regulation, illustrating the complex dynamics between political contributions and administrative policy. For years, the Food and Drug Administration has operated with a degree of autonomy intended to prioritize scientific data and public health outcomes over economic interests. However, the recent intervention by the White House in the oversight of electronic cigarettes and flavored tobacco products suggests a pivoting of priorities, where the preservation of industry revenue streams appears to have gained equal, if not greater, footing than the regulatory mandates of the FDA.
The core of this conflict lies in the aggressive stance the FDA took regarding the teen vaping epidemic, which had been characterized by health officials as a national crisis. Under the leadership of the then-commissioner, the agency prepared a series of stringent regulations designed to remove flavored nicotine products from the market—products that data suggested were specifically attractive to minors. This regulatory push was rooted in the agency’s mission to prevent a new generation from developing lifelong nicotine dependencies, a goal that would necessitate significant operational changes and financial losses for major tobacco and vaping conglomerates.
In response to these looming restrictions, the tobacco industry mobilized a sophisticated lobbying effort, leveraging long-standing financial relationships with political organizations and campaign groups. By channeling significant capital into political coffers, the industry ensured its concerns were heard at the highest levels of the executive branch. This strategic use of financial influence created a direct counter-narrative to the FDA’s scientific warnings, framing the proposed regulations as an unnecessary government overreach that would stifle small businesses and limit adult access to harm-reduction alternatives.
The resulting friction between the presidency and the FDA reached a breaking point when the administration signaled its intent to scale back or delay the proposed vaping bans. This intervention was not merely a procedural disagreement but a fundamental challenge to the authority of the FDA commissioner. By siding with industry stakeholders over the advice of specialized health experts, the administration effectively neutralized the agency’s momentum. This shift in support provided a lucrative victory for tobacco firms, who saw their stock prices stabilize and their market share protected against the immediate threat of regulatory disruption.
The resignation of the FDA commissioner serves as a stark indicator of the internal collapse of consensus within the federal government regarding health policy. When a high-ranking official exits a post in response to the overruling of scientific recommendations by political actors, it raises profound questions about the independence of regulatory bodies. The departure signaled to the scientific and medical communities that the traditional barrier between health advocacy and political fundraising had become increasingly porous, allowing economic interests to dictate the pace of public safety initiatives.
This sequence of events highlights a clear cause-and-effect relationship between political donations and legislative outcomes. The infusion of industry capital into partisan groups created an environment where the executive branch was incentivized to view tobacco regulation through an economic lens rather than a clinical one. Consequently, the dilution of the FDA’s proposed flavor ban was not an isolated policy shift but a direct result of effective industry advocacy and the administration’s receptivity to its largest financial backers.
From an analytical perspective, the “lucrative win” secured by the tobacco industry carries long-term implications for the future of nicotine consumption in the United States. By delaying the removal of flavored products, the industry has maintained its ability to recruit new users, thereby ensuring future revenue. This delay provides the industry with the necessary time to adapt its marketing strategies and litigate remaining regulations, effectively slowing the progress of public health initiatives that have been decades in the making.
Furthermore, the incident establishes a concerning precedent for other federal agencies tasked with oversight. If the FDA’s scientific expertise can be secondary to political considerations, other entities such as the Environmental Protection Agency or the Occupational Safety and Health Administration may find their mandates similarly compromised. This erosion of administrative independence suggests a move toward a more transactional form of governance, where regulatory relief is a tangible byproduct of political support.
The public health consequences of this policy pivot are likely to be observed over the coming decades. Epidemiologists warn that the window of opportunity to curb youth vaping is closing, and the delay in federal action may lead to a permanent increase in nicotine addiction rates among adolescents. While the tobacco industry celebrates a period of regulatory reprieve and financial growth, the social cost—measured in healthcare expenditures and long-term illness—may far outweigh the short-term economic benefits cited by industry proponents.
The legal and legislative battle is far from over, as public health advocacy groups and state attorneys general have begun to fill the void left by federal inaction. Several states have moved to implement their own flavor bans, creating a fragmented regulatory environment that poses new challenges for tobacco companies. However, the lack of a cohesive national strategy remains a significant advantage for the industry, as it allows for jurisdictional shopping and prolonged legal challenges against localized restrictions.
Ultimately, the victory for the tobacco industry under this administration underscores the enduring power of lobbying in Washington. It serves as a case study in how financial influence can reshape the priorities of a government, even when those priorities are ostensibly aimed at protecting the lives of citizens. The tension between profit-driven enterprise and the public good remains the defining conflict of modern regulatory policy, with the tobacco industry’s latest win marking a decisive chapter in that ongoing struggle.
As the political landscape continues to evolve, the degree to which health agencies can reclaim their autonomy will depend on future legislative reforms and the prevailing political climate. For now, the tobacco industry remains in a position of strength, having demonstrated that its political alliances are a formidable defense against the mandates of scientific authority. The long-term impact on the FDA’s credibility and the nation’s health will be the true measure of this controversial alignment between the boardroom and the oval office.